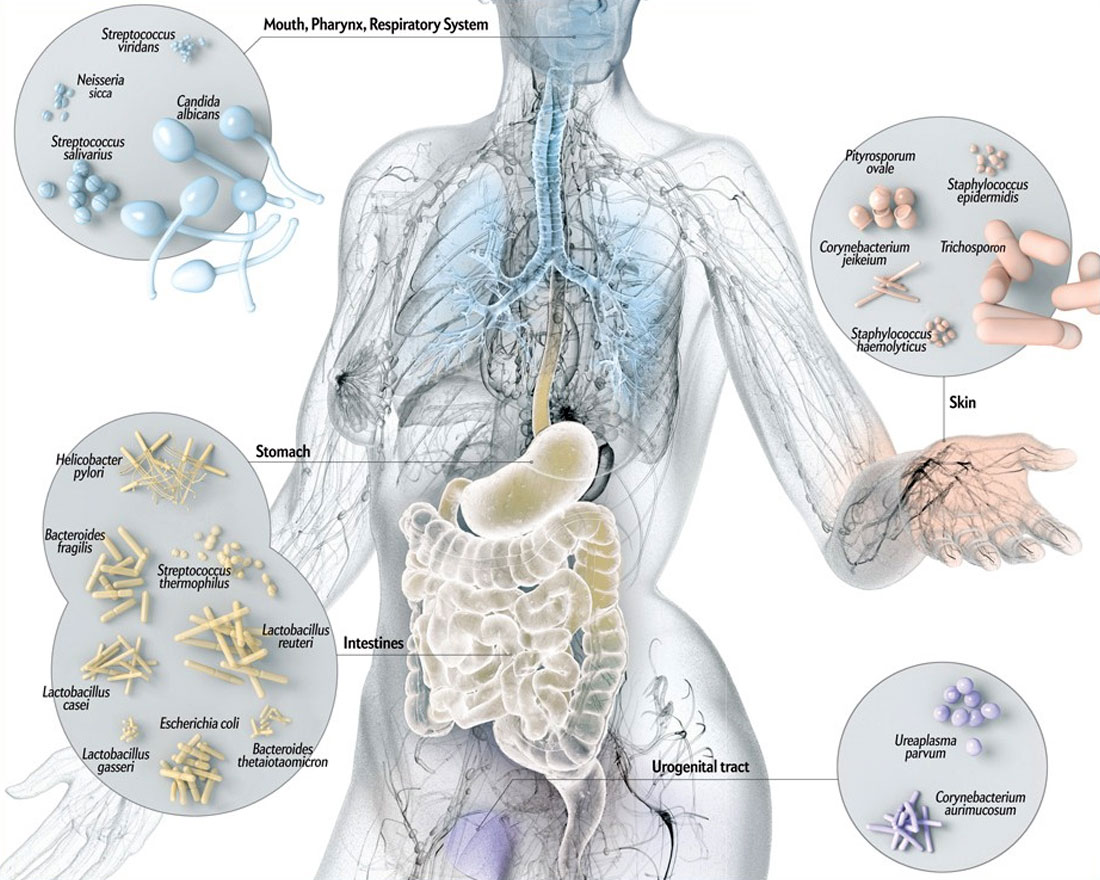
Growing up, I viewed the human body as a castle, with defenses to keep all the nasty bacteria and viruses out. White blood cells were the soldiers, sent off to do battle at breaches in the walls. In fairy tales everything good is inside the castle walls, everything bad outside; however, in the real world it’s often hard to distinguish between good and evil, and all sorts are found on both sides of the castle walls. Lately medical research is showing that far from being a single organism, the human body is full of microorganisms connected by complex and little understood interdependencies (Figure 1). As the saying goes, no man is an island, and perhaps we’d be better to compare each human to a coral reef teeming with a rich range of microscopic life, rather than a castle or island. In fact, similar to a coral reef, increasing evidence is suggesting that a compromised human microbiota could be a factor in a number of diseases / medical conditions, often in ways that are not immediately obvious, because the interdependencies between the human body and its many resident microorganisms are complex.
Bacterial colonies are found in many areas of the body, and the relationships between the host body and its bacteria can be divided into four categories. In a commensal relationship one organism benefits and the other is unaffected; with a parasitic relationship one organism benefits to the detriment of the other; mutualism occurs when both organisms benefit; finally, with amensalism one organism is harmed with the other unaffected. Until recently it was believed that the relationships between the human body and its microbiota were simple, and mostly parasitic or commensal. Now an understanding is forming that in fact there are many mutualistic relationships, and many of the relationships change depending on circumstances, or are a mix with some benefits and some harms coming to both human and bacteria.
Of course, it has long been known that if numbers of bacteria get too high, or if bacteria get into parts of the body that they aren’t normally found in, then bacterial infections will result. Sepsis, pneumonia and peritonitis are the terms given to bacterial infections in the blood, lungs and stomach cavity, and these and all the other types of infection are serious threats to human health. But only about 1% of bacteria can cause infection, and the roles that all these bacteria play in a healthy human’s functions are very interesting.
Let’s get the terminology out of the way. The collection of microorganisms in a human body is known as the microbiota, and includes bacteria, fungi, and archaea (prokaryotes – single celled organisms with no membrane-bound cell components such as nuclei). The term human microbiome refers to the human microbiota’s genomes. Note that an organism’s genome is its genetic material, essentially its DNA, and RNA in the case of RNA viruses. The apparent emphasis on the human microbiota’s genomes is simply explained by the observation that the microorganisms in question are hard to distinguish from one another, and analysis of their DNA is the most effective way of doing this.
In 2012 the Human Microbiome Project Consortium identified and catalogued many of the microorganisms found in the human body using advanced DNA sequencing methods, a huge and rewarding effort. It was long known that most of these organisms live in colonies that use enzymes to break down sugars/ carbohydrates in order to survive. But this study revealed that because the types, locations, and levels of carbohydrates vary considerably from human to human, so too do the types and numbers of microorganisms. So we all have our own unique landscape of microscopic life within us, primarily found in our skin and mucus membranes, and it is constantly changing as each bacterial colony moves towards an equilibrium with its dynamic environment. The sheer number and variety of microorganisms living within each of us is astounding. Wexler (2007) states that by numbers of cells we are 10% human and 90% bacterial, and by number of genes we are 1% human, 99% bacterial!
Some of the useful things these bacteria do for us include:
- Aid digestion, e.g. break down lactose and proteins
- Synthesize valuable vitamins
- Aid the immune system
- Help fight cancer cells
- Keep levels of harmful bacteria low
Instead of trying to cover all angles of this topic, I will focus on a few interesting examples of bacteria that live inside and on us.
Digestive System
The digestive system is where most of the human microbiota are found, and where they do some of their most important work. The stomach is too acidic for most bacteria, so very low levels of bacteria are found here, and the same for the small intestine with its relatively high residual acidity. It is in the large intestine where a large variety (500 – 1000) and huge numbers of bacteria are found, and we know that they play an important role in a healthy digestive system. These are mostly anaerobic bacteria, and a lot of the carbohydrates they break down are ones we could not digest without their help.
“Though people can (barely) survive with no gut flora, the microorganisms perform a host of useful functions, such as fermenting unused energy substrates, training the immune system, preventing growth of harmful species, regulating the development of the gut, producing vitamins for the host (such as biotin and vitamin K), and producing hormones to direct the host to store fats.” (Wikimedia Foundation, Inc., 2016). Low levels of the most common gut bacteria, Faecalibacterium prausnitzii, are directly related to Crohn’s disease, obesity and even asthma.
Researchers spend a lot of time analyzing human poop, since it’s such a convenient place to find the gut microbiota – about 60% of dry feces is bacterial.
Skin Microbiota
Most of the bacteria living in and on skin tissue depend on the products excreted by various skin-related glands such as sebum from the sebaceous glands, and fatty and amino acids produced by sweat glands. The body has evolved such that it depends on bacteria to break down these substances to a certain extent in order to keep the glands and pores clear and in working condition. Certain issues can result when the numbers and types of bacteria get out of balance, for example acne. Other normal human characteristics are actually a direct result of bacterial involvement, for example it is bacterial action that creates the smell of sweat; the sweat is otherwise odorless. An extremely interesting finding is that the types and levels of diversity in human skin microbiota partly determine how attractive a person is to mosquitos; this would help explain why some people get eaten alive while the mosquitos seem to avoid others.
The Mouth
I liken the bacteria in our mouths to lichen (couldn’t resist), algae and other plants clinging to rocks in mountain streams. In order to survive the bacteria have to stay there – being washed down into the stomach means a certain death in a cauldron of acids. So they have developed ways to cling to our teeth and gums, primarily through the formation of plaque, essentially an adhesive biofilm, and of course they also nestle in to the nooks and crannies between our teeth and gums. The reward is an otherwise ideal bacterial environment – it’s moist with an abundant and regular stream of nutrients going by.
Contrary to many of the bacteria previously mentioned, most of the oral bacteria are harmful, and there is an ongoing battle between our oral defense systems and the colonies of plaque-forming bacteria. It’s really like the Wild West, a dangerous frontier that the bacteria hope to colonize. Various types of Streptococci are prevalent, and it is S. mutans that usually establishes the first foothold, with other types of bacteria following it. S. mutans is able to produce a sticky, dextran-based polysaccharide from sucrose that forms the basis for plaque. A byproduct of this is lactic acid, and this creates pits and fissures in the tooth enamel, allowing other bacteria to take hold. All this of course can lead to tooth decay and gum disease. In older times teeth lasted long enough for the much shorter human life spans, but now keeping our teeth healthy into old age is a struggle.
Reproductive Organs
Women used to wage an ongoing battle against supposedly harmful germs and bacteria in their lower reproductive tracts; besides homegrown methods involving vinegar, there was an entire section in the drug store selling disinfectant and deodorizing weaponry. Today we know all of that was actually harmful. Several types of bacteria are an important component of a healthy vaginal ecosystem, most notably the genus Lactobacillus which produces lactic acid and hydrogen peroxide, both of which guard against infection. Removing these bacteria actually increases the risk of infection and other undesirable conditions. The feminist wars have been fought in numerous battles big and small, but moving from a time when disinfection was seen as a requirement, to today where a very pretty term – vaginal flora – describes the female microbiota, must be taken as a victory for feminism!
In the spirit of gender equality, how about the male microflora? Semen usually contains several varieties of bacteria along with sperm, but they are usually picked up along the urinary tract. The most common are Lactobacillus, Pseudomonas, Prevotella, and Gardnerella. A recent study suggests that Lactobacillus is a key companion of healthy sperm and may help reduce levels of Pseudomonas and Prevotella which are associated with unhealthy sperm (Weng, et al., 2014). The foreskin area is a topic unto itself. An unhygienic foreskin creates a perfect environment for bacterial infection, but over washing, especially with soap, can also cause problems. Interestingly, circumcision and when it is done actually seems to change the types of bacteria present in the area. Overall, the situation is similar within the male and female reproductive systems – bacteria are present, and certain ones play positive roles, but others are present as well and if the balance is upset there can be negative effects.
Immune System
There are known examples of resident bacteria assisting our immune system. A good example is Bacteroides fragilis, which is found in our colon. B. fragilis produces a molecule known as polysaccharide A (PSA), which human dendritic immune cells pick up and present to undifferentiated T cells. The PSA acts as a trigger, causing the T cells to convert to regulatory T cells that help reduce the effects of inflammatory T cells. The relationship between us and B. fragilis was thought to be commensal, but this T cell interaction shows it to be a bit more than that. Further, if B. fragilis gets out of its colon home and into the bloodstream or nearby tissues it can be quite dangerous – it is involved in ~90% of anaerobic peritoneal infections (Wexler, 2007). This serves as a useful example of the complexity of the relationships between humans and their microbiota. On the one hand we have evolved to depend on them, on the other hand given the right circumstances, whether it’s a weakened immune system, a physical injury, changes caused by medication, diet or whatever, they can quickly cause or be involved in infections, disease and other situations dangerous to our health.











Share This Column